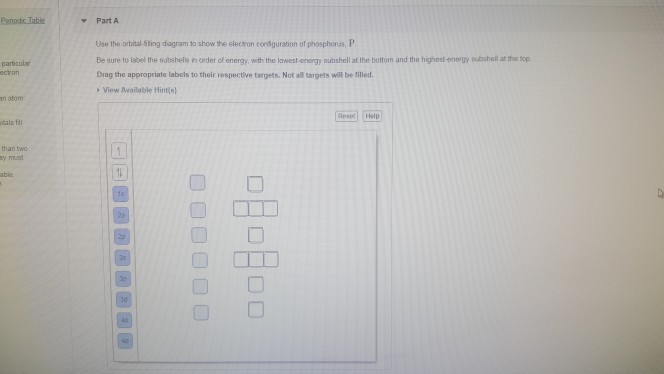
Use The Orbital Filling Diagram To Show The Electron Configuration Of Phosphorus P
Be sure to arrange the subshells in order of e. The p orbital can hold up to six electrons.
 Electron Configuration Revolvy
Electron Configuration Revolvy
Not all targets will be filled.
Use the orbital filling diagram to show the electron configuration of phosphorus p. Use the periodic table to identify the neutral atoms having the following electron configurations. Note that the last term in the phosphorus electron configuration will be 1s2 2s2 2p6 3s2 3p3. The 3p electrons are in three different orbitals and have the same spin.
In this video well use the electron configuration chart to help us write the notation for phosphorus. Because the atomic number is equivalent to the number of electrons the standard electron configuration can be used to draw the orbital diagram. With an atomic number of 15 two electrons.
Show transcribed image text use the orbital filling diagram to show the electron configuration of phosphorus p. Stack the subshells in order of energy with the lowest energy subshell at the bottom and the highest energy subshell at the top. Since 1s can only hold two electrons the next 2 electrons for phosphorous go in the 2s orbital.
Show the electron configuration of phosphorusp. Give the complete ground state electron configuration for silicon si. Stack the subshells in order of energy with the lowest energy subshell at the bottom and the highest energy subshell at the top.
Stack the subshells in order of energy with the lowest energy subshell at the bottom and the highest energy subshell at the top. Show the orbital filling diagram for br bromine. Be sure to label the subshells in order of energy with the lowest energy subshell at the bottom and the highest energy drag the appropriate labels to their respective targets.
In writing the electron configuration for phosphorus the first two electrons will go in the 1s orbital. The next six electrons will go in the 2p orbital. Be sure to arrange the subshells in order of energy with the lowest energy subshell at the bottom and the highest energy subshell at the top.
Show the electron configuration of phosphorusp. Show the orbital filling diagram for n nitrogen. The orbital diagram for phosphorus consists of five electrons in the third shell eight in the second and two in the first shell closest to the nucleus.
Electron configuration element ne 3s 2 magnesium ar 4s 2 3d 5 manganese kr 5s 2 4d 10 5p 3 antimony 5.
Quantum Numbers And Electron Configurations
 Ground State Electron Configuration Definition Example Video
Ground State Electron Configuration Definition Example Video
 Electron Configuration Wikipedia
Electron Configuration Wikipedia
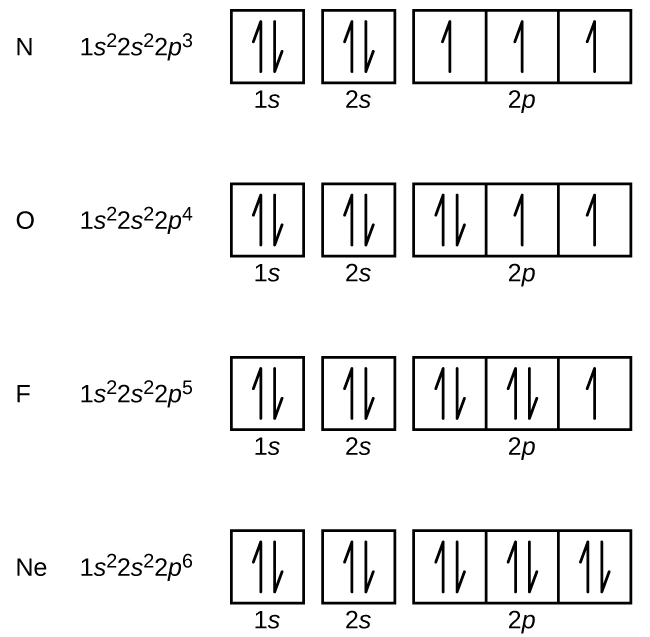 6 4 Electronic Structure Of Atoms Electron Configurations Chemistry
6 4 Electronic Structure Of Atoms Electron Configurations Chemistry
 Electron Configuration Orbital Noble Gas Electron Configuration
Electron Configuration Orbital Noble Gas Electron Configuration
Chapter 7 Quantum Theory And The Electronic Structure Of Atoms
 Ground State Electron Configuration Definition Example Video
Ground State Electron Configuration Definition Example Video

 Molecular Orbital Diagram Wikipedia
Molecular Orbital Diagram Wikipedia
3 2 Electron Configurations Of Atoms
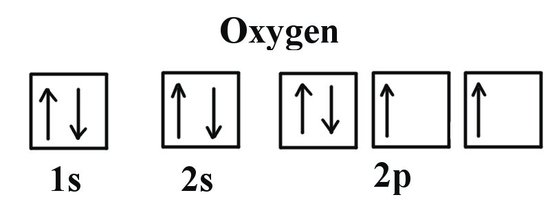 1 4 Electron Configurations Electronic Orbital Diagrams Review
1 4 Electron Configurations Electronic Orbital Diagrams Review
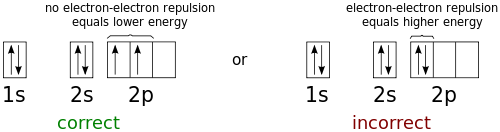 High School Chemistry Orbital Configurations Wikibooks Open Books
High School Chemistry Orbital Configurations Wikibooks Open Books
 Period 4 Subshell Electronic Configuration
Period 4 Subshell Electronic Configuration
 How Are Electrons Distributed In Different Orbits Electronic
How Are Electrons Distributed In Different Orbits Electronic

 How To Draw The Lewis Dot Structure For P 3 Phosphide Ion Youtube
How To Draw The Lewis Dot Structure For P 3 Phosphide Ion Youtube
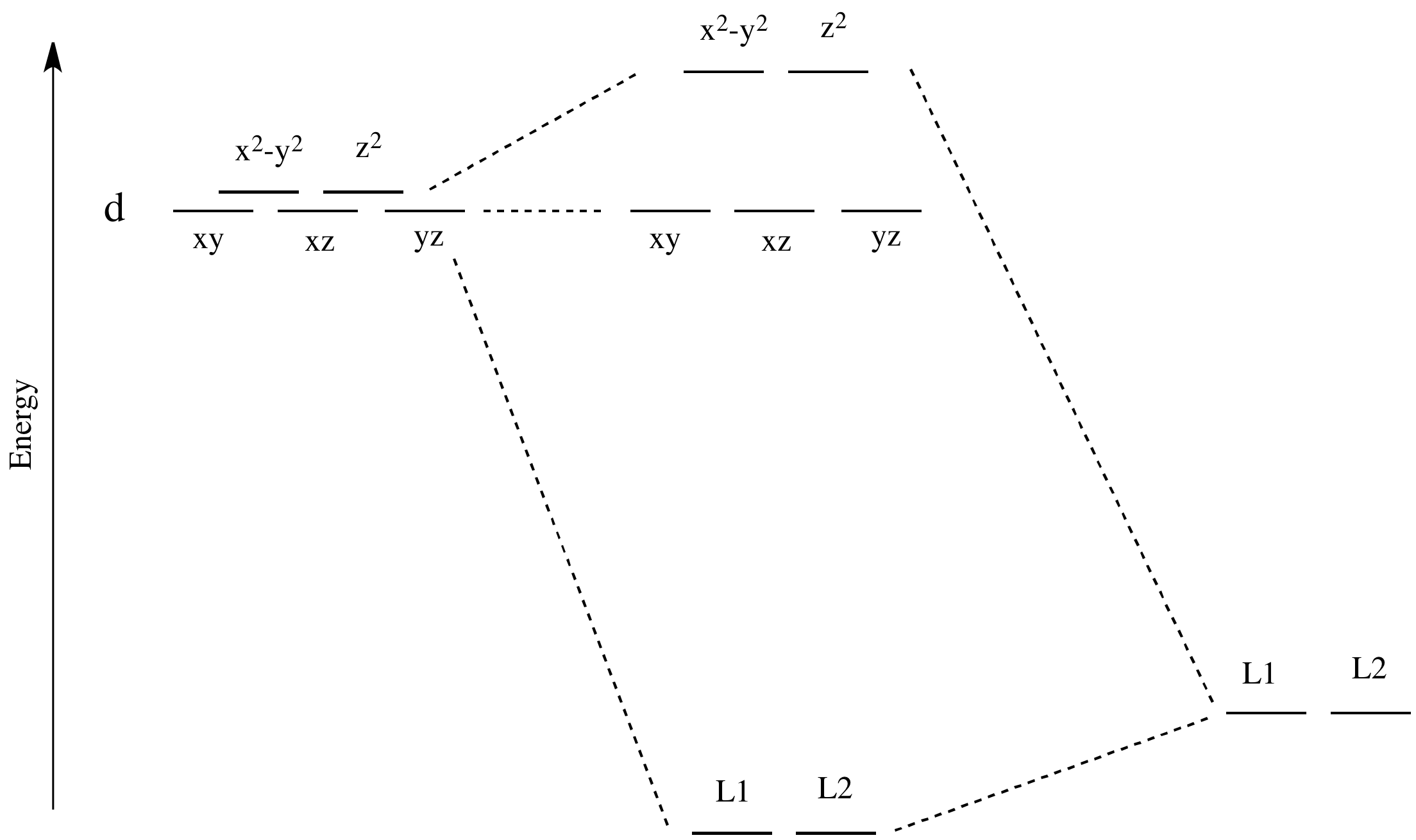
0 Response to "Use The Orbital Filling Diagram To Show The Electron Configuration Of Phosphorus P"
Post a Comment