Which Diagram Shows Electrons Violating The Pauli Exclusion Principle
Which set of orbital diagrams shows a violation of the pauli exclusion principle. Full s orbital shields the electron in the p orbital from the nucleus.
 Pdf Vip An Experiment To Search For A Violation Of The Pauli
Pdf Vip An Experiment To Search For A Violation Of The Pauli
Which one of the following represents an incorrect set of quantum numbers for an electron in an atom arranged as n l m l and m s.

Which diagram shows electrons violating the pauli exclusion principle. To show the electron configuration for an atom what is the advantage of using an orbital notation compared to a dot structure. Atomic radii of group 13 elements are generally bigger. Fermions include elementary particles such as quarks the constituent particles of protons and neutrons electrons and neutrinos.
What is the maximum number of electrons that can occupy the subshell 3d. Thus it can be seen that in option c electrons in last 2 subshell have electrons with same spin which is a violation of pauli exclusion principle. The electron configuration for helium he is shown below.
Chemistry chapter 2 quiz 6. How many electrons can the n 4 shell hold. When we draw electrons we use up and down arrows.
The pauli exclusion principle governs the behavior of all fermions particles with half integer spin while bosons particles with integer spin are not subject to it. Which diagram shows electrons violating the pauli exclusion principle. 1s2 which diagram shows the correct distribution of electrons in the electron shells of a helium atom.
Based on these rules. Pauli violation because the two electrons in the 3s have the same spin. Aufbau principle electrons fill orbitals starting at the lowest available energy state before filling higher states 1s before 2s.
Orbital notation shows the spin of the electrons. One should be pointing down. Electrons in group 13 elements are closer to the nucleus.
An orbital can hold 0 1 or 2 electrons only and if there are two electrons in the orbital they must have opposite paired spins. Recognizing examples and non examples of the aufbau principle drawing electron configurations with aufbauorbital diagram finding violations of pauli exclusion principle hunds rule and aufbau principle in diagrams identifying elements that are paramagnetic or diamagnetic recognizing excited versus ground states. Paulis exclusion principle says that no two electrons can have the same quantum numbers so the maximum electrons that can occupy each orbital are 2 and they much have opposite spins.
Not mc014 4jpgmc014 5jpgmc014 6jpg up down up down up down. Further an orbital can contain a maximum of only two electrons the two electrons must have opposing spins.
 Solved Shown Below Are Portions Of Orbital Diagrams Repre Sen
Solved Shown Below Are Portions Of Orbital Diagrams Repre Sen
 All The Past Results From Pep Violation Tests For Electrons With A
All The Past Results From Pep Violation Tests For Electrons With A
This Lecture Deals With Atomic And Nuclear Structure
 Experimental Search For The Violation Of Pauli Exclusion Principle
Experimental Search For The Violation Of Pauli Exclusion Principle
 Solved The P Subshells Of An Atom Have Three P Orbitalis
Solved The P Subshells Of An Atom Have Three P Orbitalis
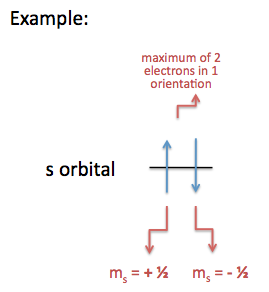 12 10 Electron Spin And The Pauli Principle Chemistry Libretexts
12 10 Electron Spin And The Pauli Principle Chemistry Libretexts
 The Pauli Exclusion Principle Physics
The Pauli Exclusion Principle Physics
 Homework 8 Electron Configuration Ppt Video Online Download
Homework 8 Electron Configuration Ppt Video Online Download
 Solved The P Subshells Of An Atom Have Three P Orbitals T
Solved The P Subshells Of An Atom Have Three P Orbitals T
New Limits On Bosonic Dark Matter Solar Axions Pauli Exclusion
Test Of The Pauli Exclusion Principle In The Vip 2 Underground
 Solved Four Possible Electron Configurations For A Nitrog
Solved Four Possible Electron Configurations For A Nitrog
 New Experimental Limit On The Pauli Exclusion Principle Violation By
New Experimental Limit On The Pauli Exclusion Principle Violation By
 New Experimental Limit On The Pauli Exclusion Principle Violation By
New Experimental Limit On The Pauli Exclusion Principle Violation By
Search For Pauli Exclusion Principle Violating Atomic Transitions
 Does Neutron Star Collapse Violate Quantum Theory Plancktime
Does Neutron Star Collapse Violate Quantum Theory Plancktime
Pauli Aufbau Hund Chemistnate Lessons
 Aufbau Diagram For Calcium Wiring Schematic Diagram
Aufbau Diagram For Calcium Wiring Schematic Diagram
Exchange Antisymmetry And Pauli Repulsion
 Electronic Configurations Ppt Download
Electronic Configurations Ppt Download
0 Response to "Which Diagram Shows Electrons Violating The Pauli Exclusion Principle"
Post a Comment